Fda Prior Notice Form

One to support the registration of facilities that manufacture process pack or hold food products intended for consumption in the united states and one to receive prior notice before.
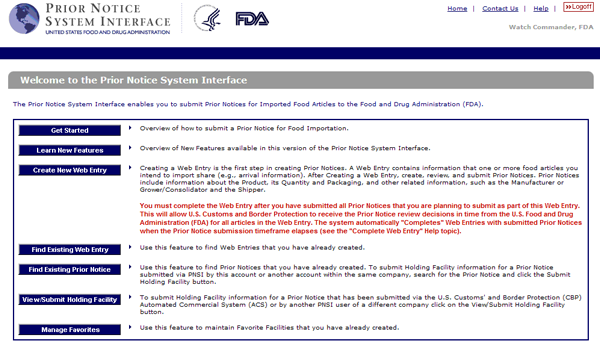
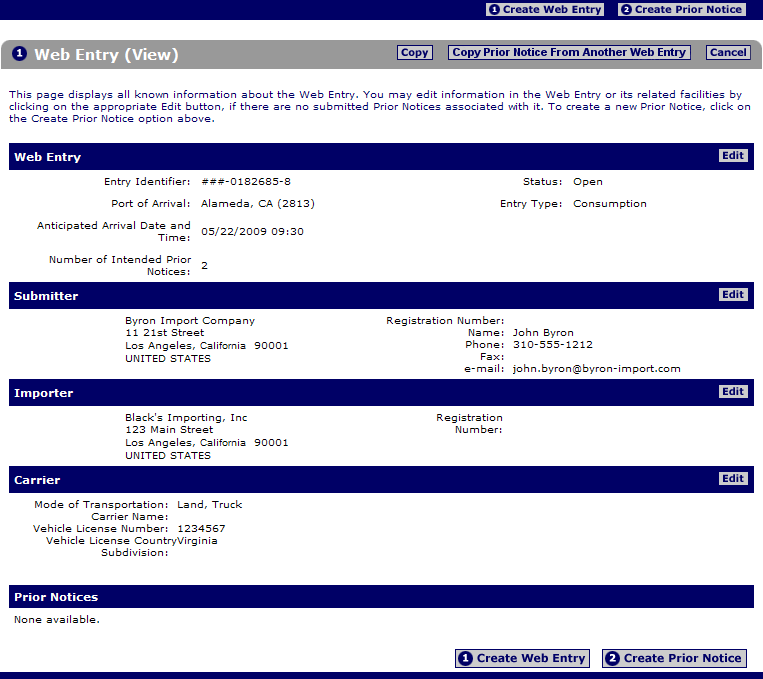
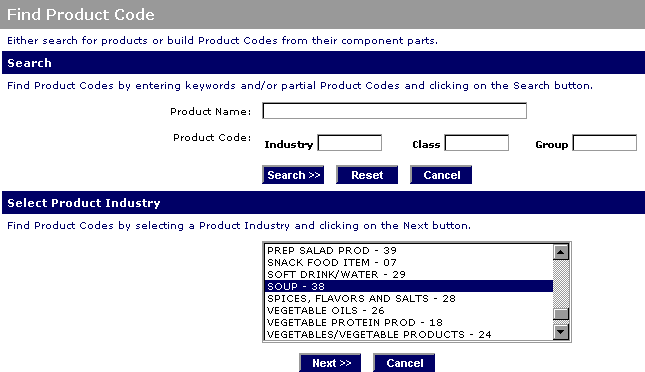
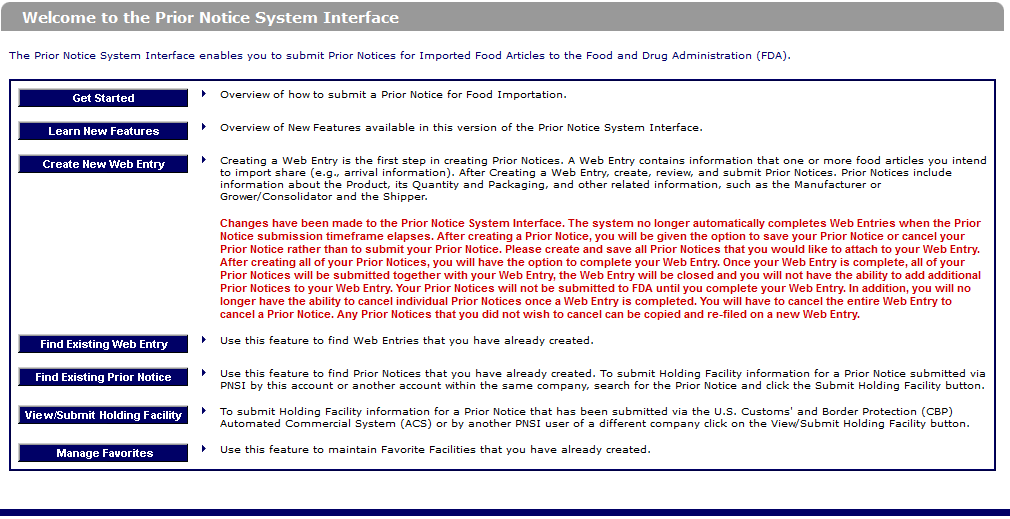
Fda prior notice form. By submitting the form you agree and warrant that the information stated above are accurate truthful and current and authorize liberty management group ltd. Fda quickly and properly. Food supply from outside the country through the prior notice of imported foods. Introduction to prior notice requirements for imported foods.
Under the bioterrorism act of 2002 fda regulates foods entering the u s. Are food and food products. A final rule prior notice rule in the federal register requiring submission to fda of prior notice of food including food for animals that is imported or offered for import into the united. To submit the prior notice information to the u s.
Fda requires the filing of prior notice for all food beverage and dietary supplements shipments entering the united states. As of december 12 2003 fda must be notified in advance of any shipments of food for humans and other animals that are imported into the u s unless. Fis was created in part in response to the bioterrorism act of 2002 which gave high priority to improved information management to help protect the food supply the act requires that fda develop two systems. Contact fda follow fda on facebook follow fda on twitter view fda videos on youtube subscribe to fda rss feeds fda homepage contact number 1 888 info fda 1 888 463 6332.
In 2002 congress passed the bioterrorism act as a part of its. Submitter name job title submitter e mail. Upon receipt of prior notice fda will issue a confirmation number in the form of a bar code that must accompany most food shipments. Nearly 20 of all imports into the u s.
Food and drug administration u s. About prior notice prior notice requirement.